Recently, the EndoFresh single-use digestive endoscopy system successfully obtained certification from the Saudi Food and Drug Authority (SFDA), enabling the full product portfolio to officially enter the market of Saudi Arabia.
Even more exciting, the EndoFresh 300 Series successfully completed the world’s first clinical ESG procedure (Endoscopic Sleeve Gastroplasty) performed using a single-use digestive endoscope in Saudi Arabia.This milestone marks the official entry of China-developed single-use digestive endoscopy technology into the international high-end therapeutic field.
Chinese Innovation Gains Global Recognition Once Again
EndoFresh Opens a New Chapter in Global Expansion
As one of the most influential healthcare markets in the Middle East, Saudi Arabia maintains extremely strict standards for medical device market access.
Leveraging innovative technology and outstanding performance, EndoFresh single-use digestive endoscopes successfully passed the review of the Saudi Food and Drug Authority and obtained market entry approval.

Looking back to April this year, EndoFresh appeared at an academic conference jointly organized by the Saudi Gastroenterology Association (SGA) and the King Faisal Specialist Hospital & Research Centre.
During the conference, the EndoFresh single-use digestive endoscopy system became a focal product and performed impressively across multiple sessions:
Served as the dedicated teaching device during the PEG hands-on workshop
Demonstrated high-definition visualization and precise maneuverability during live demonstrations
Its ready-to-use and single-use design gained recognition from experts across multiple medical institutions
Following the approval of the 200 Series diagnostic digestive endoscopes in Saudi Arabia earlier this January, the 300 Series therapeutic digestive endoscopes have also recently received certification, laying a solid foundation for EndoFresh’s continued expansion in the Saudi market.
Comprehensive Product Portfolio
Meeting Diverse Clinical Needs
200 Series
The 200 Series is designed by EndoFresh as a single-use digestive endoscope dedicated to diagnostic procedures.
Meets the needs of routine endoscopic examinations
Equipped with a 3.2 mm working channel, enabling basic therapeutic procedures
300 Series
The 300 Series is a comprehensive upgrade built upon the 200 Series, optimized specifically for therapeutic endoscopic procedures:
Larger working channels and expanded angulation designed for complex therapeutic procedures
Multiple length options available for treating lesions in different anatomical locations
Applicable for hemostasis, foreign body removal, mucosal resection/dissection, tunneling techniques, variceal treatment, NOTES procedures, dual-endoscope operations, and bariatric endoscopic therapies
The World’s First ESG Procedure Using a Single-Use Endoscope
EndoFresh 300 Series Demonstrates Outstanding Performance
The world’s first ESG procedure (Endoscopic Sleeve Gastroplasty) performed using a single-use digestive endoscope in Saudi Arabia represents a significant milestone.
Endoscopic Sleeve Gastroplasty (ESG) is an advanced weight-loss procedure that reduces stomach volume through full-thickness endoscopic suturing techniques.
It offers significant advantages such as minimal trauma, faster recovery, and fewer complications, providing a safer and more effective minimally invasive treatment option for patients with obesity.
The procedure was completed successfully, and the EndoFresh 300 Series single-use digestive endoscope delivered excellent performance.
The physicians involved in the surgery highly commented:
”ESG is among the most complex therapeutic endoscopic interventions. What impressed me most was how user-friendly the 300 Series scope felt, offering:
Smooth and agile manipulation;
Excellent image quality for precise targeting.
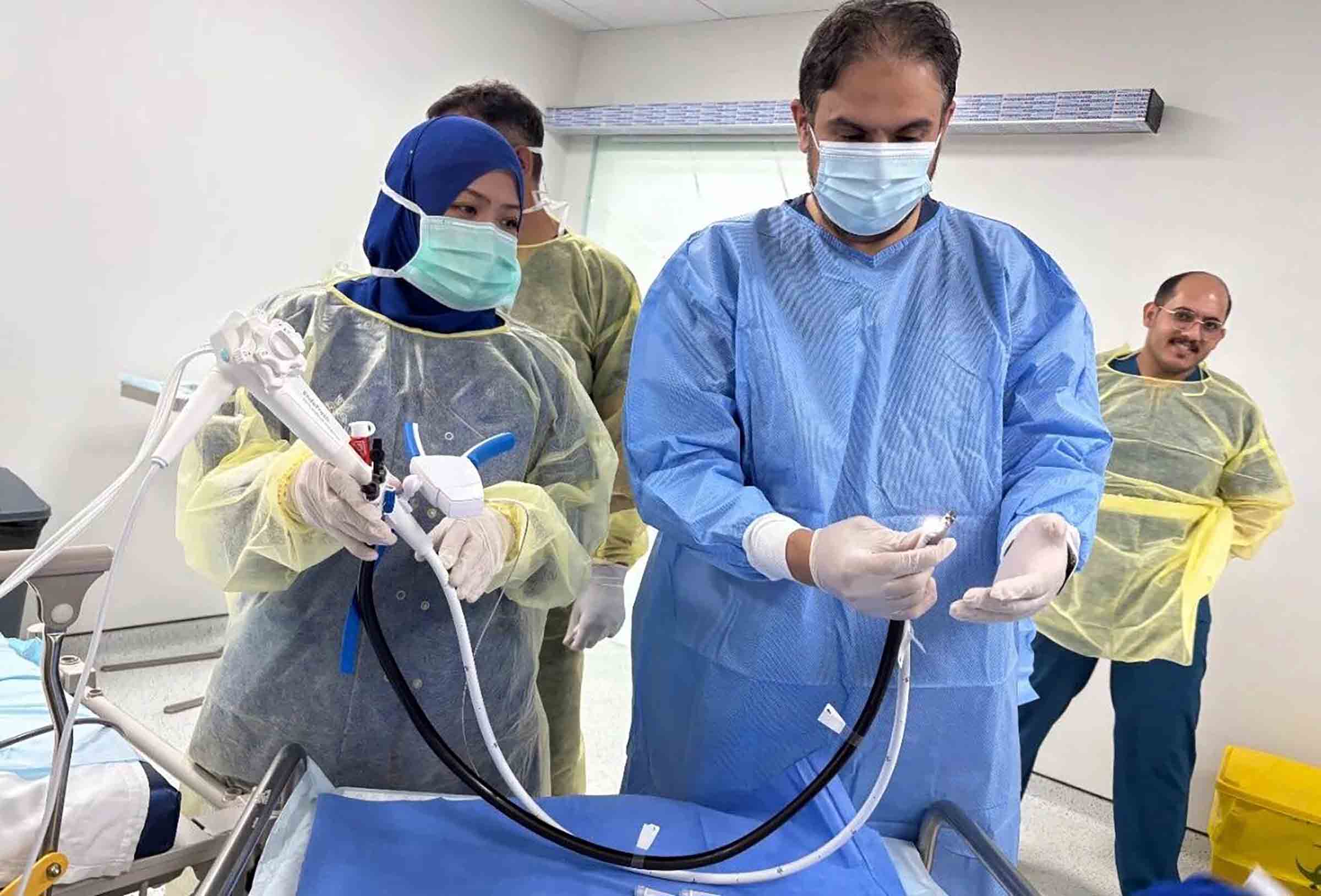
The success of this procedure not only verified the technical feasibility of using single-use endoscopy in such complex procedures but also laid the groundwork for broader clinical applications.
It represents another milestone for EndoFresh as it advances into the field of endoscopic bariatric surgery.
Expanding in the Global Market
Leading Industry Development Through Innovation
With the successful Saudi certification and ESG procedure, EndoFresh once again demonstrates the strength of China’s medical technology innovation.
Looking ahead, EndoFresh will continue to adhere to an innovation-driven strategy, continuously improving product performance and providing global healthcare institutions with higher-quality single-use digestive endoscopy solutions, enabling more patients to benefit from safe and efficient endoscopic diagnosis and treatment.
Let the world witness the power of China’s innovation in medical technology!
